






 ften images taken for medical purposes look similar; one easily recognizes that they show the same part of human anatomy. However, the information contained in them as well as their spatial resolution is different. Some depict only anatomy, others mix anatomy with metabolic information, as the four images of a brain in Figure 02-01 prove.
ften images taken for medical purposes look similar; one easily recognizes that they show the same part of human anatomy. However, the information contained in them as well as their spatial resolution is different. Some depict only anatomy, others mix anatomy with metabolic information, as the four images of a brain in Figure 02-01 prove.
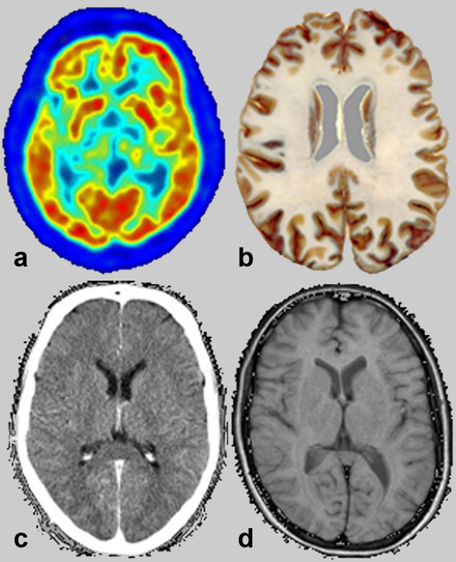
Figure 02-01:
Somehow similar pictures: transverse slices through the brain. The methods of creation vary, from (a) positron emmission tomography, (b) an anatomical specimen, (c) x-ray computed tomography, and (d) magnetic resonance imaging. The main fundamental discrepancy between the different techniques is explained on Table 02-01.
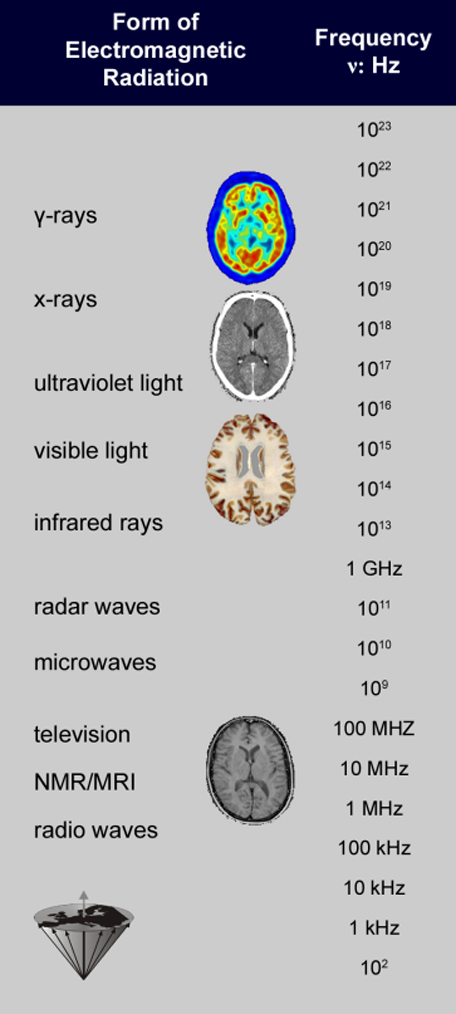
Table 02-01:
Spectrum of electromagnetic radiation. The radiation used for NMR and MR imaging is several times lower than that for γ-rays and x-rays.
Matter consists of atoms (e.g, ¹H, ¹²C, ¹⁶O, ³¹P, etc.). An atom of one element differs from an atom of another element in its internal structure: the number of protons, neutrons, and electrons. Protons and neutrons comprise the nucleus; protons are positively charged, neutrons possess no charge, and the electrons orbiting around the nucleus are negatively charged. Different nuclear compositions and numbers of surrounding electrons are reflected in different physical properties.
Unlike color or texture some physical properties are not easily perceived. Such is the case with the magnetic properties of the nuclei which are the basis of the nuclear magnetic resonance phenomenon. Although these properties are not easily visualized, they are well defined and obey certain rules.
This allows us to make analogies to more well-known phenomena so as to aid our understanding.
With the help of electromagnetic (radio) waves, the magnetic properties permit the production of images of the human body which can furnish information about morphology and function of the human organism. Owing to the stupendous spread of frequencies and wavelengths of electromagnetic waves, their interaction with matter is very dissimilar in the different parts of the spectrum.
The radiation used for magnetic resonance imaging is quite different from x-ray and γ-radiation (Table 02-01). It stretches from AM frequencies through mobile, amateur radio and TV to FM radio frequencies, is approximately nine orders of magnitude smaller than the frequencies corresponding to x- or γ-rays (used for radioisotope examinations), and is considered biologically safe (more in Chapter 18).
Since Röntgen’s discovery of the x-rays more than a century has passed. He succeeded in generating images of the human body with x-rays which result from interactions between these rays and the electron clouds of atoms.
Nuclear magnetic resonance signals stem from the interaction of radiowaves with the atomic nuclei themselves. This is the reason for the completely different imaging equipment necessary and the different contrast behavior of magnetic resonance imaging as compared with other medical imaging techniques.